See Figure 15.10 for an illustration of osmotic movement of fluid out of a cell when hypertonic IV fluid is administered due to a higher concentration of solutes (pink molecules) in the bloodstream compared to the cell. 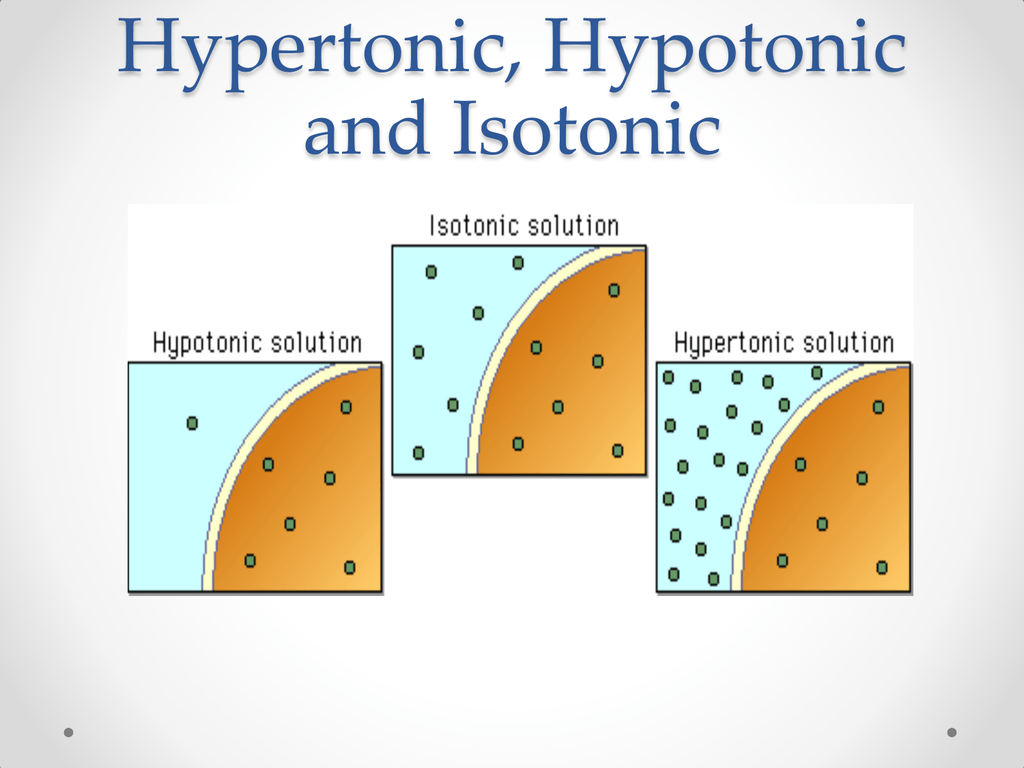
This causes the osmotic movement of water out of the cells and into the intravascular space to dilute the solutes in the blood. When infused, hypertonic fluids cause an increased concentration of dissolved solutes in the intravascular space compared to the cells. An example of hypertonic IV solution is 3% Normal Saline (3% NaCl). Hypertonic solutions have a higher concentration of dissolved particles than blood. Figure 15.9 Hypotonic IV Solution Causing Osmotic Movement of Fluid Into Cell Therefore, patient status should be monitored carefully when hypotonic solutions are infused. It is also possible to cause worsening hypovolemia and hypotension if too much fluid moves out of the intravascular space and into the cells. However, if too much fluid moves out of the intravascular compartment into cells, cerebral edema can occur. See Figure 15.9 for an illustration of the osmotic movement of fluid into a cell when a hypotonic IV solution is administered, causing lower concentration of solutes (pink molecules) in the bloodstream compared to within the cell. For this reason, hypotonic fluids are used to treat cellular dehydration. This imbalance causes osmotic movement of water from the intravascular compartment into the intracellular space. When hypotonic IV solutions are infused, it results in a decreased concentration of dissolved solutes in the blood as compared to the intracellular space. An example of a hypotonic IV solution is 0.45% Normal Saline (0.45% NaCl). Hypotonic solutions have a lower concentration of dissolved solutes than blood.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
